Delivering Exceptional Patient Satisfaction^
94%
Satisfaction with overall vision without glasses.7
96%
Satisfaction with reading on a smartphone or tablet.8
92%
Satisfaction with ability to see steps and curbs at night.8


Satisfaction with overall vision without glasses.7
Satisfaction with reading on a smartphone or tablet.8
Satisfaction with ability to see steps and curbs at night.8
Freeform diffractive profile contributes to a low incidence of bothersome visual disturbances.9
93% reported no or mild halos, glare, or starbursts at one month post-op.9
Values rounded to the nearest 1%
Retrospective, multi-center, real-world clinical analysis of reported outcomes at 1 month post-operative visit, n=96. Symptoms reported without a specified severity level were classified as mild in the chart above.
A continuous, full range of vision with better near compared to PanOptix®.*†
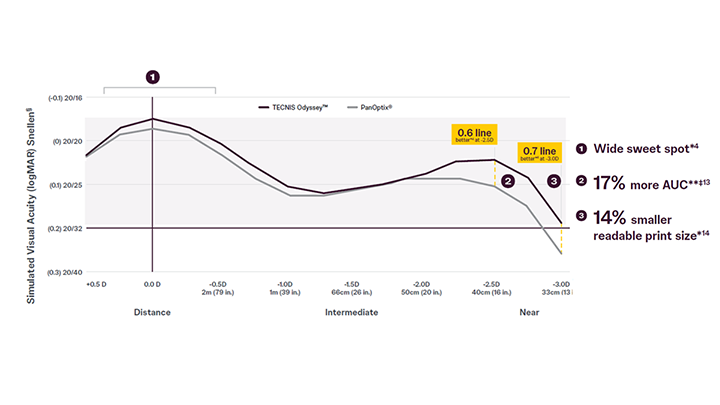
*Based on bench testing compared to PanOptix®
**AUC=Area under the curve
† continuous 20/25 or better
‡above 0.2 LogMar (~20/32 Snellen) compared to PanOptix®
§Snellen VA was converted from logMAR VA. A Snellen notation of 20/20-2 or better indicates a logMAR VA of 0.04 or better
Superior low-light contrast compared to PanOptix®. ^^^5,11,12

*Based on pre-clinical bench testing (white light MTF at 50 c/mm measured for 5mm pupil in the ACE model)
Best-in-class compares to competitor IOLs of comparable range of vision.
*According to ISO 11979-7:2024, based on the clinical study of the parent IOL
¶Compared to PanOptix® based on bench testing and head-to-head clinical studies of parent lens
^^ Compared to PanOptix® based on bench testing (white light MFT at 50 c/mm measured for 3mm & 5mm pupil in the ACE model)
†Compared to TECNIS SYNERGY™ based on bench testing (white light MFT at 50 c/mm measured for 3mm and 5mm pupil in the ACE model)
**Continuous 20/25 or better based on pre-clinical bench testing for TECNIS Odyssey™
^ Values rounded to the nearest 1%. Based on 3-month postoperative data from a multicenter, observational clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33)
$ Compared to TECNIS SYNERGY™ and TECNIS™ Multifocal IOLs based on pre-clinical bench testing
*** Compared to TECNIS SYNERGY™ based on bench testing
^^^Compared to leading competitor full range of vision IOLs based on bench testing (white light MTF at 50 c/mm measured for 3mm & 5mm pupil in the ACE model)
1. Data on File. 2024DOF4002 (prospective, multicenter, randomized, three-way-masked clinical study comparing subjects bilaterally implanted with TECNIS Synergy™ IOL (n=132) vs TECNIS™ 1-Piece Monofocal IOL (n=131) at 6-months post-op)
2. Data on File. 2024DOF4003
3. Data on File. 2024DOF4005
4. Data on File. DOF2023CT4023
5. Data on File. DOF2023CT4007
6. Data on File. DOF2019OTH4002
7. Data on File. 2024DOF4027 (ambispective, multicenter, observational clinical study evaluating subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33) at 3-months post-op)
8. Data on File. 2024DOF4029 (Based on 3-month postoperative data from a multicenter, observational clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33))
9. Data on File. DOF2023CT4050 (Based on 1-month postoperative data from a multicenter, retrospective, real-world clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=96))
10. Dick, H. Burkhard MD, et. al. (2022 November). A Comparative Clinical Evaluation of a New TECNIS Presbyopia Correcting Intraocular Lens Against a Trifocal Intraocular Lens (“Forte 1”)
11. Data on File. 2024DOF4033
12. Data on File. DOF2018OTH4004
13. Data on File. 2024DOF4015
14. Data on File. DOF2023CT4056
15. Data on File. DOF2020CT4014 - Forte 1: A ComparativeClinical Evaluation of a New TECNIS® Presbyopia Correcting Intraocular Lens Against a PanOptix® Intraocular Lens - DEFOCUS CURVES AND VISUAL ACUITY RESULTS.
INDICATIONS AND IMPORTANT SAFETY INFORMATION FOR TECNIS ODYSSEY™ IOL WITH TECNIS SIMPLICITY™ DELIVERY SYSTEM, MODEL DRN00V AND TECNIS ODYSSEY™ TORIC II IOL WITH TECNIS SIMPLICITY™ DELIVERY SYSTEM, MODELS DRT150, DRT225, DRT300, DRT375
Rx Only
INDICATIONS FOR USE:
The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS Odyssey™ IOL, which is indicated for primary implantation for the visual correction of aphakia in adult patients, with less than 1 diopter of pre-existing corneal astigmatism, in whom a cataractous lens has been removed. The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS Odyssey™ Toric II IOLs that are indicated for primary implantation for the visual correction of aphakia and for reduction of refractive astigmatism in adult patients with greater than or equal to 1 diopter of preoperative corneal astigmatism, in whom a cataractous lens has been removed. Compared to an aspheric monofocal lens, the TECNIS Odyssey™ IOLs mitigate the effects of presbyopia by providing improved visual acuity at intermediate and near distances to reduce eyeglass wear, while maintaining comparable distance visual acuity. The lens is intended for capsular bag placement only.
PRECAUTIONS
Careful preoperative evaluation and sound clinical judgment should be used by the surgeon to decide the benefit/risk ratio before implanting a lens in a patient with one or more of these conditions.
Before Surgery
• Pupil abnormalities
• Prior corneal refractive or intraocular surgery
• Choroidal hemorrhage
• Chronic severe uveitis
• Concomitant severe eye disease
• Extremely shallow anterior chamber
• Medically uncontrolled glaucoma
• Microphthalmos
• Non-age-related cataract
• Proliferative diabetic retinopathy (severe)
• Severe corneal dystrophy
• Severe optic nerve atrophy
• Irregular corneal astigmatism
• Amblyopia
• Macular disease
• Pregnancy
During Surgery
• Excessive vitreous loss
• Non-circular capsulotomy/capsulorhexis
• The presence of radial tears known or suspected at the time of surgery
• Situations in which the integrity of the circular capsulotomy/capsulorhexis cannot be confirmed by direct visualization
• Cataract extraction by techniques other than phacoemulsification or liquefaction
• Capsular rupture
• Significant anterior chamber hyphema
• Uncontrollable positive intraocular pressure
• Zonular damage
Potential complications generally associated with cataract surgery include, but are not limited to: endophthalmitis/intraocular infection, hypopyon, hyphema, IOL dislocation, persistent cystoid macular edema, pupillary block, retinal detachment/tear, persistent corneal stromal edema, persistent uveitis, persistent raised intraocular pressure (IOP) requiring treatment (e.g., AC tap), retained lens material, or toxic anterior segment syndrome, or any other adverse event that leads to permanent visual impairment or requires surgical or medical intervention to prevent permanent visual impairment. Adverse events that may be associated with use of the device include: IOL dislocation, tilt or decentration, visual symptoms requiring lens removal, residual refractive error, secondary surgical intervention (including IOL repositioning or removal).
25. Do not leave the lens in a folded position more than 10 minutes.
26. When the delivery system is used improperly, the lens may not be delivered properly, (i.e., haptics may be broken). Please refer to the specific instructions for use provided.
WARNINGS