Delivering Exceptional Patient Satisfaction^
94%
Satisfaction with overall vision without glasses.7
96%
Satisfaction with reading on a smartphone or tablet.8
92%
Satisfaction with ability to see steps and curbs at night.8


Satisfaction with overall vision without glasses.7
Satisfaction with reading on a smartphone or tablet.8
Satisfaction with ability to see steps and curbs at night.8
Freeform diffractive profile contributes to a low incidence of bothersome visual disturbances.9
93% reported no or mild halos, glare, or starbursts at one month post-op.9
Values rounded to the nearest 1%
Retrospective, multi-center, real-world clinical analysis of reported outcomes at 1 month post-operative visit, n=96. Symptoms reported without a specified severity level were classified as mild in the chart above.
A continuous, full range of vision with better near compared to PanOptix®.*†
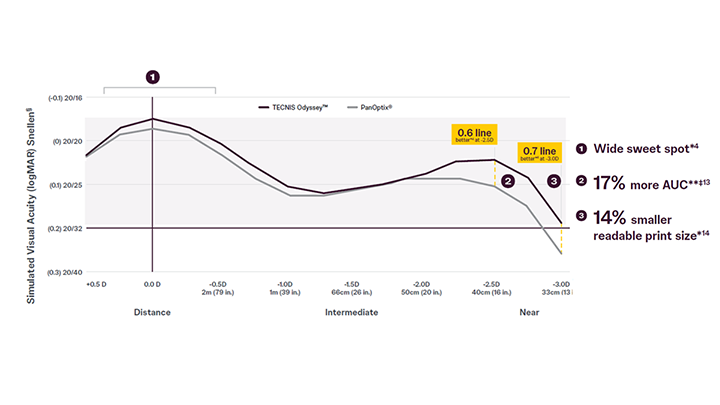
*Based on bench testing compared to PanOptix®
**AUC=Area under the curve
† continuous 20/25 or better
‡above 0.2 LogMar (~20/32 Snellen) compared to PanOptix®
§Snellen VA was converted from logMAR VA. A Snellen notation of 20/20-2 or better indicates a logMAR VA of 0.04 or better
Superior low-light contrast compared to PanOptix®. ^^^5,11,12

*Based on pre-clinical bench testing (white light MTF at 50 c/mm measured for 5mm pupil in the ACE model)
Best-in-class compares to competitor IOLs of comparable range of vision.
Learn more about the evolution of the TECNIS™ portfolio over the last 25 years!
*According to ISO 11979-7:2024, based on the clinical study of the parent IOL
¶Compared to PanOptix® based on bench testing and head-to-head clinical studies of parent lens
^^ Compared to PanOptix® based on bench testing (white light MFT at 50 c/mm measured for 3mm & 5mm pupil in the ACE model)
†Compared to TECNIS SYNERGY™ based on bench testing (white light MFT at 50 c/mm measured for 3mm and 5mm pupil in the ACE model)
**Continuous 20/25 or better based on pre-clinical bench testing for TECNIS Odyssey™
^ Values rounded to the nearest 1%. Based on 3-month postoperative data from a multicenter, observational clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33)
$ Compared to TECNIS SYNERGY™ and TECNIS™ Multifocal IOLs based on pre-clinical bench testing
*** Compared to TECNIS SYNERGY™ based on bench testing
^^^Compared to leading competitor full range of vision IOLs based on bench testing (white light MTF at 50 c/mm measured for 3mm & 5mm pupil in the ACE model)
1. Data on File. 2024DOF4002 (prospective, multicenter, randomized, three-way-masked clinical study comparing subjects bilaterally implanted with TECNIS Synergy™ IOL (n=132) vs TECNIS™ 1-Piece Monofocal IOL (n=131) at 6-months post-op)
2. Data on File. 2024DOF4003
3. Data on File. 2024DOF4005
4. Data on File. DOF2023CT4023
5. Data on File. DOF2023CT4007
6. Data on File. DOF2019OTH4002
7. Data on File. 2024DOF4027 (ambispective, multicenter, observational clinical study evaluating subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33) at 3-months post-op)
8. Data on File. 2024DOF4029 (Based on 3-month postoperative data from a multicenter, observational clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=33))
9. Data on File. DOF2023CT4050 (Based on 1-month postoperative data from a multicenter, retrospective, real-world clinical study in the U.S. evaluating visual and patient-reported outcomes from subjects bilaterally implanted with TECNIS Odyssey™ IOL (n=96))
10. Dick, H. Burkhard MD, et. al. (2022 November). A Comparative Clinical Evaluation of a New TECNIS Presbyopia Correcting Intraocular Lens Against a Trifocal Intraocular Lens (“Forte 1”)
11. Data on File. 2024DOF4033
12. Data on File. DOF2018OTH4004
13. Data on File. 2024DOF4015
14. Data on File. DOF2023CT4056
15. Data on File. DOF2020CT4014 - Forte 1: A ComparativeClinical Evaluation of a New TECNIS® Presbyopia Correcting Intraocular Lens Against a PanOptix® Intraocular Lens - DEFOCUS CURVES AND VISUAL ACUITY RESULTS.
INDICATIONS AND IMPORTANT SAFETY INFORMATION FOR TECNIS ODYSSEY™ IOL WITH TECNIS SIMPLICITY™ DELIVERY SYSTEM, MODEL DRN00V AND TECNIS ODYSSEY™ TORIC II IOL WITH TECNIS SIMPLICITY™ DELIVERY SYSTEM, MODELS DRT150, DRT225, DRT300, DRT375
Rx Only
INDICATIONS FOR USE:
The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS ODYSSEY™ IOL, which is indicated for primary implantation for the visual correction of aphakia in adult patients, with less than 1 diopter of pre-existing corneal astigmatism, in whom a cataractous lens has been removed. The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS ODYSSEY™ Toric II IOLs that are indicated for primary implantation for the visual correction of aphakia and for reduction of refractive astigmatism in adult patients with greater than or equal to 1 diopter of preoperative corneal astigmatism, in whom a cataractous lens has been removed. Compared to an aspheric monofocal lens, the TECNIS ODYSSEY™ IOLs mitigate the effects of presbyopia by providing improved visual acuity at intermediate and near distances to reduce eyeglass wear, while maintaining comparable distance visual acuity. The lens is intended for capsular bag placement only.
PRECAUTIONS
1.This is a single use device. Do not resterilize the lens or the delivery system. Most sterilizers are not equipped to sterilize the soft acrylic material and the preloaded inserter material without producing undesirable side effects.
2.Do not store the device in direct sunlight or at a temperature under 41°F (5°C) or over 95°F (35°C).
3.Do not autoclave the delivery system.
4.Do not advance the lens unless ready for lens implantation.
5.The contents are sterile unless the package is opened or damaged.
6.The recommended temperature for implanting the lens is at least 63°F (17°C).
7.The use of balanced salt solution or ophthalmic viscosurgical devices (OVDs), is required when using the delivery system. For optimal performance when using OVD, use the HEALON™ family of OVDs. The use of balanced salt solution with additives has not been studied for this product.
8.Do not use if the delivery system has been dropped or if any part was inadvertently struck while outside the shipping box. The sterility of the delivery system and/ or the lens may have been compromised.
9.When performing refraction in patients implanted with the lens, interpret results with caution when using autorefractors or wavefront aberrometers that utilize infrared light, or when performing a duochrome test. Confirmation of refraction with maximum plus manifest refraction technique is strongly recommended.
10.The ability to perform some eye treatments (e.g., retinal photocoagulation) may be affected by the IOL optical design.
11.Recent contact lens usage may affect the patient’s refraction; therefore, in contact lens wearers, surgeons should establish corneal stability without contact lenses prior to determining IOL power.
12.The surgeon should target emmetropia as this lens is designed for optimum visual performance when emmetropia is achieved.
13.Care should be taken to achieve centration of the intraocular lens in the capsular bag.
14.Prior to surgery, the surgeon must inform prospective patients of the possible risks and benefits associated with the use of the device and provide them a copy of the patient information brochure.
15.Children under the age of 2 years are not suitable candidates for intraocular lenses.
16.The lens should not be placed in the ciliary sulcus.
17.Carefully remove all viscoelastic and do not over-inflate the capsular bag at the end of the case. Residual viscoelastic and/or over-inflation of the capsular bag may allow the lens to rotate, causing misalignment of the toric lens with the intended axis of placement.
18.The TECNIS™ Toric IOL Calculator includes a feature that accounts for posterior corneal astigmatism (PCA). The PCA is based on an algorithm that combines published literature (Koch, et al., 2012) and a retrospective analysis of data from a TECNIS™ Toric multi-center clinical study. The PCA algorithm for the selection of appropriate cylinder power and axis of implantation was not assessed in the prospective TECNIS™ Toric IOL U.S. IDE study and may yield results different from those in the TECNIS ODYSSEY™ Toric II IOL labeling. Please refer to the TECNIS™ Toric IOL Calculator user manual for more information.
19.The use of methods other than the TECNIS™ Toric IOL Calculator to select cylinder power and appropriate axis of implantation were not assessed in the TECNIS™ Toric IOL U.S. IDE study and may not yield similar results. Accurate keratometry and biometry, in addition to the use of the TECNIS™ Toric IOL Calculator (www.TecnisToricCalc.com) are recommended to achieve optimal visual outcomes for the toric lens.
20.All preoperative surgical parameters are important when choosing a toric lens for implantation, including preoperative keratometric cylinder (magnitude and axis), incision location, the surgeon's estimated surgically induced astigmatism (SIA) and biometry. Variability in any of the preoperative measurements can influence patient outcomes and the effectiveness of treating eyes with lower amounts of preoperative corneal astigmatism. The effectiveness of TECNIS ODYSSEY™ Toric II IOLs in reducing postoperative residual astigmatism in patients with preoperative corneal astigmatism <1.0 diopter has not been demonstrated.
21.Patients with a predicted postoperative astigmatism greater than 1.0 D may not be suitable candidates for implantation with the TECNIS ODYSSEY™ and TECNIS ODYSSEY™ Toric II IOLs, as they may not obtain the benefits of reduced spectacle wear or improved intermediate and near vision seen in patients with lower astigmatism.
22.All corneal incisions were placed temporally in the TECNIS™ Toric IOL U.S. IDE study. If the surgeon chooses to place the incision at a different location, outcomes may be different from those obtained for the TECNIS™ Toric IOL. Note that the TECNIS™ Toric IOL Calculator incorporates the surgeon’s estimated SIA and incision location when providing IOL options.
23.Do not reuse.
24.The safety and effectiveness of the TECNIS ODYSSEY™ IOL and the TECNIS ODYSSEY™ Toric II IOL have not been substantiated in patients under the age of 22 or those with preexisting ocular conditions and intraoperative complications, including those specified in the Warnings and Precautions, such as pupil abnormalities, prior corneal refractive or intraocular surgery, acute or chronic ophthalmic diseases or conditions (see below for examples).
Careful preoperative evaluation and sound clinical judgment should be used by the surgeon to decide the benefit/risk ratio before implanting a lens in a patient with one or more of these conditions.
Before Surgery
During Surgery
Potential complications generally associated with cataract surgery include, but are not limited to: endophthalmitis/intraocular infection, hypopyon, hyphema, IOL dislocation, persistent cystoid macular edema, pupillary block, retinal detachment/tear, persistent corneal stromal edema, persistent uveitis, persistent raised intraocular pressure (IOP) requiring treatment (e.g., AC tap), retained lens material, or toxic anterior segment syndrome, or any other adverse event that leads to permanent visual impairment or requires surgical or medical intervention to prevent permanent visual impairment. Adverse events that may be associated with use of the device include: IOL dislocation, tilt or decentration, visual symptoms requiring lens removal, residual refractive error, secondary surgical intervention (including IOL repositioning or removal).
25.Do not leave the lens in a folded position more than 10 minutes.
26.When the delivery system is used improperly, the lens may not be delivered properly, (i.e., haptics may be broken). Please refer to the specific instructions for use provided.
WARNINGS
Patients should have well-defined visual needs and be informed of possible visual effects (such as a perception of halo, starbursts or glare around lights), which may be expected in nighttime or poor visibility conditions. Patients may perceive these visual effects as bothersome, which, on rare occasions, may be significant enough for the patient to request removal of the IOL.