*Overall spectacle wear at 6 months, none/a little/some of the time
**Prospective, randomized, multi-center 6-month clinical study. N=66


Of patients occasionally or never wore glasses*1
Of patients would recommend the lens to friends or relatives**2
Of patients reported no very bothersome visual disturbances1
*Overall spectacle wear at 6 months, none/a little/some of the time
**Prospective, randomized, multi-center 6-month clinical study. N=66
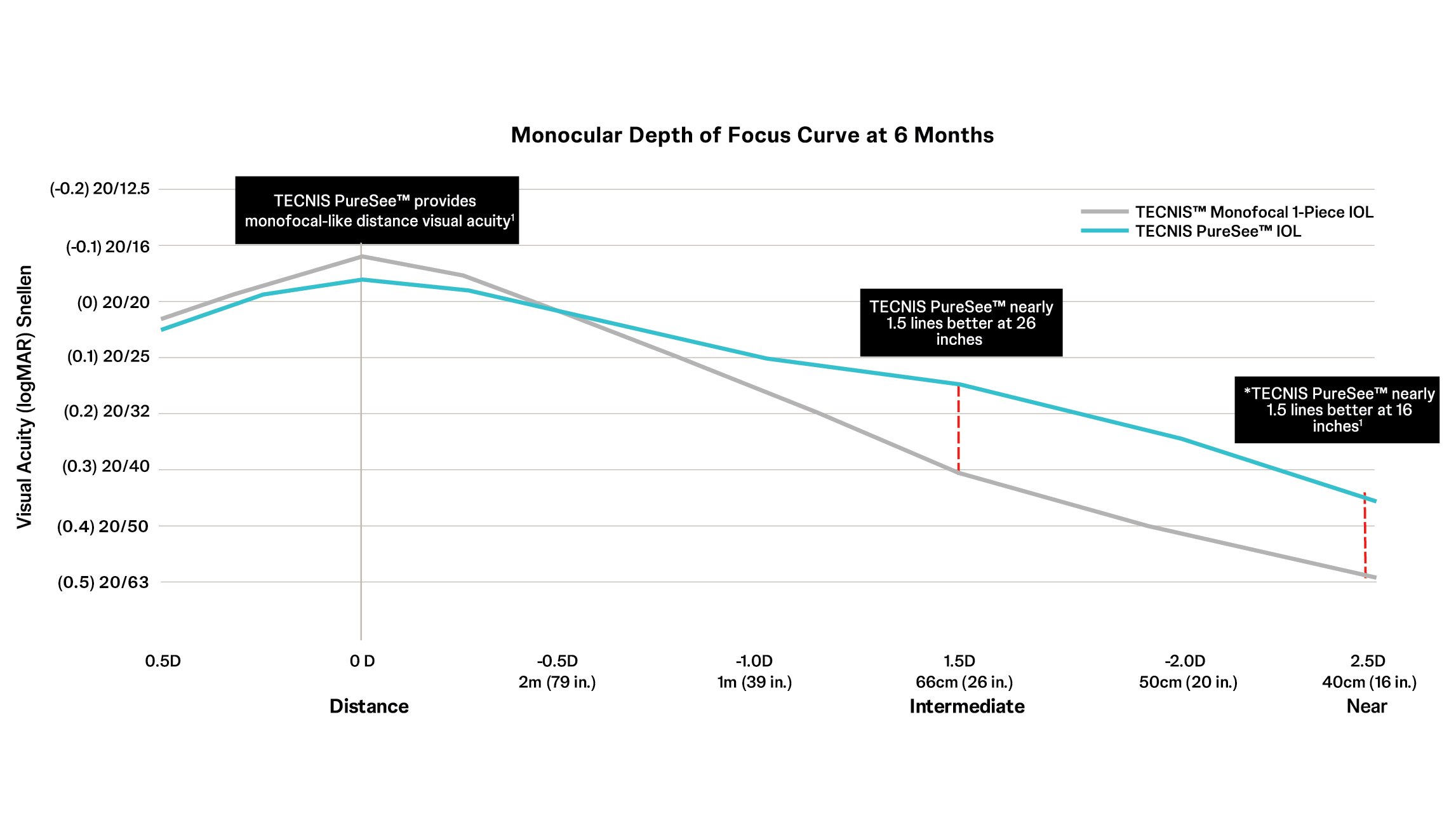
TECNIS PureSee™ delivers monofocal-like distance visual acuity1
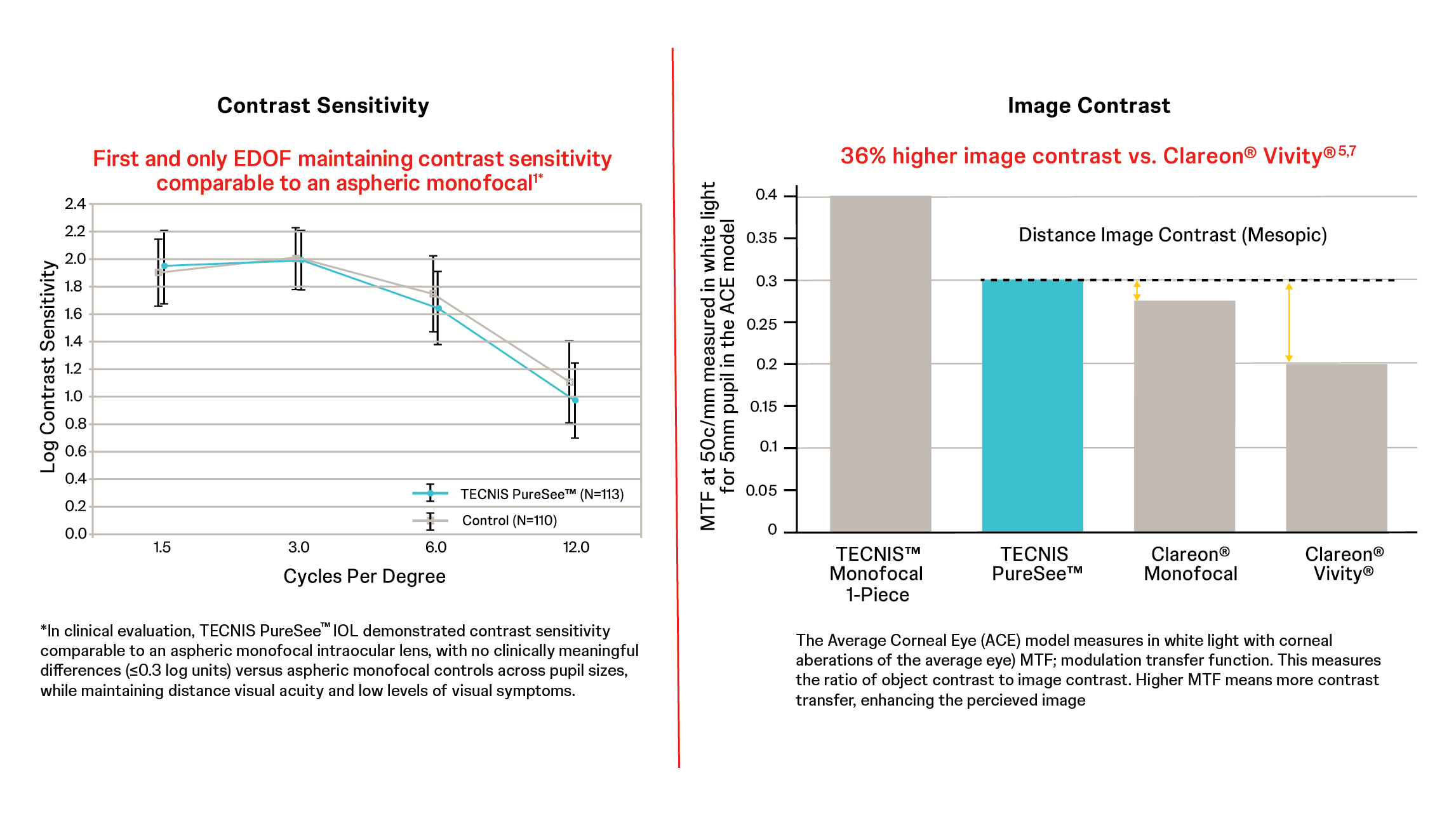
Even under defocus, leading to high patient satisfaction3
![TECNIS[^TM] Monofocal 1-Piece IOL TECNIS[^TM] Monofocal 1-Piece IOL](https://images.contentstack.io/v3/assets/bltc6e7ece114c6734f/blt9fd66b3c471512a1/693770ce07a64267d5e495ed/TECNIS_Monofocal_1_piece.jpg?branch=prod_alias)
![TECNIS PureSee[^TM] IOL TECNIS PureSee[^TM] IOL](https://images.contentstack.io/v3/assets/bltc6e7ece114c6734f/bltda59093306dae652/693770ce6c564f5093cdb319/TECNIS_PureSee.jpg?branch=prod_alias)
optical bench testing: DOF2023CT4012
![TECNIS[^TM] Monofocal 1-Piece IOL and TECNIS PureSee[^TM] TECNIS[^TM] Monofocal 1-Piece IOL and TECNIS PureSee[^TM]](https://images.contentstack.io/v3/assets/bltc6e7ece114c6734f/blt6d81330bf5ed26c8/69b878b056a5aef405b831a5/Tolerance-to-Refractive_13.png?branch=prod_alias)
1. TECNIS PureSee™ IOL, Model DEN00V, DFU US, Z312075E rev B.
2. DOF2023CT4043_Clinical Investigation of the TECNIS™ Intraocular Lens, Models CV1000 and CV2000. Patient satisfaction outcomes. Prospective, randomized, multi-center 6-month clinical study. N=66
3. Black DA, et al. Tolerance to refractive error with a new extended depth of focus intraocular lens. Eye (Lond). 2024;38(Suppl 1):15-20. doi: 10.1038/s41433 -024 - 03040 -1. Erratum in: Eye (Lond). 2025;39(1):203. doi: 10.1038/s41433 -024 -03422 -5.
4. 2024REF4215 Clareon® Vivity® Directions for Use.
5. DOF2018OTH4004 TECNIS Enhance™ and competitors simulated MTF.
6. 2024REF4933 Alarcon, Aixa, et al. Optical and clinical simulated performance of a new refractive extended depth of focus intraocular lens. Eye 38.Suppl 1 (2024): 4-8.
7. DOF2023CT4025.
INDICATIONS and IMPORTANT SAFETY INFORMATION for TECNIS PureSee™ IOL and TECNIS PureSee™ Toric II IOLs with TECNIS SIMPLICITY™ Delivery System
Rx Only
INDICATIONS FOR USE:
The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS PureSee™ IOL, which is indicated for primary implantation for the visual correction of aphakia in adult patients with less than 1 diopter of pre-existing corneal astigmatism in whom a cataractous lens has been removed. The lens mitigates the effects of presbyopia by providing an extended depth of focus. Compared to an aspheric monofocal IOL, the TECNIS PureSee™ IOL provides improved intermediate visual acuity, while maintaining comparable distance visual acuity. The lens is intended for capsular bag placement only.
The TECNIS SIMPLICITY™ Delivery System is used to fold and assist in inserting the TECNIS PureSee™ Toric II IOLs, which are indicated for primary implantation for the visual correction of aphakia and for reduction of refractive astigmatism in adult patients with greater than or equal to 1 diopter of preoperative corneal astigmatism in whom a cataractous lens has been removed. The lenses mitigate the effects of presbyopia by providing an extended depth of focus. Compared to an aspheric monofocal IOL, the TECNIS PureSee™ Toric II IOLs provide improved intermediate visual acuity, while maintaining comparable distance visual acuity. The lenses are intended for capsular bag placement only.
WARNINGS:
PRECAUTIONS:
Before Surgery
During Surgery
24. Potential complications generally associated with cataract surgery include, but are not limited to: endophthalmitis/intraocular infection, hypopyon, hyphema, IOL dislocation, persistent cystoid macular edema, pupillary block, retinal detachment/tear, persistent corneal stromal edema, persistent uveitis, persistent raised intraocular pressure (IOP) requiring treatment (e.g., AC tap), retained lens material, or toxic anterior segment syndrome, or any other adverse event that leads to permanent visual impairment or requires surgical or medical intervention to prevent permanent visual impairment.
ATTENTION
Reference the Directions for Use for a complete listing of Indications and Important Safety Information.