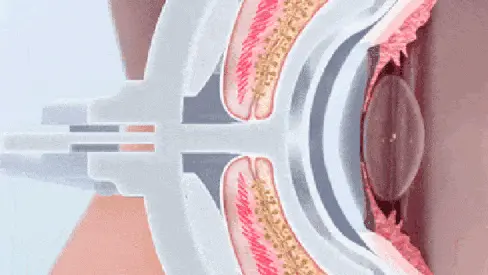
Patient Messaging
Did You Know?
- In one study, up to 86% of dry eye patients had signs of MGD3
- Approximately 1 in 5 patients with MGD are asymptomatic4
- TearScience™ LipiFlow™ has been shown to increase gland secretion threefold, on average, with just one treatment2
- Safety and long-term effectiveness have been demonstrated in peer-reviewed studies5,6,7
- TearScience™ LipiFlow™ installations are evenly divided between ophthalmology and optometry clinics
- TearScience™ LipiFlow™ is available in more than 1500 practice locations in the United States and Canada as of January 2023